 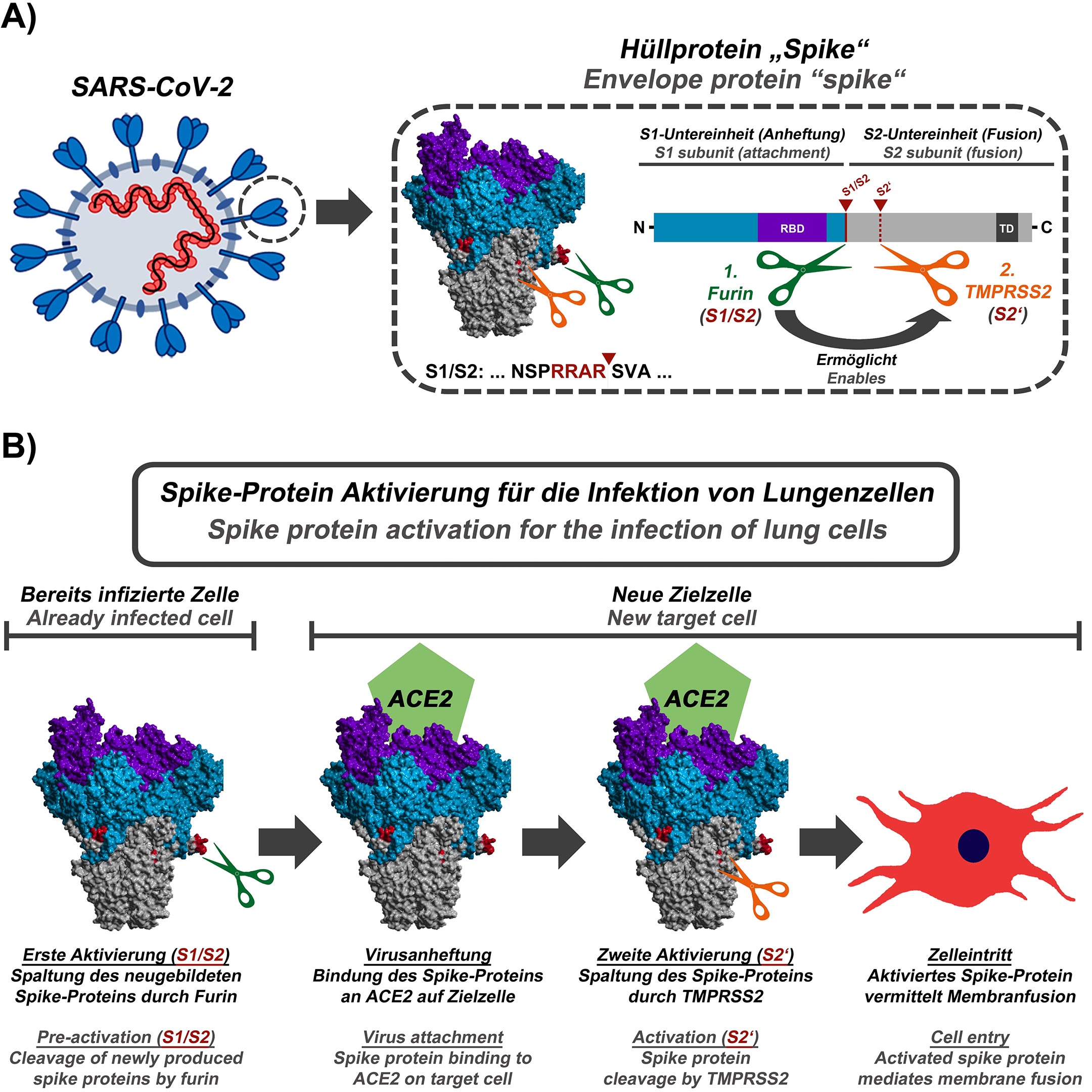
For endothelial cells of blood vessel linings, for example, the disparity between 28,000 SA-tipped CD147 receptors and 175 ACE2 receptors per cell provides a supporting indication of the role of glycans in widespread endothelial damage reported in COVID-19 patients. These glycoconjugates are abundantly distributed on the surfaces of host cells such as red blood cells (RBCs), platelets and endothelial cells and confer an associated negative electrostatic potential, additionally facilitating attachments by positively charged SARS-CoV-2 spike protein prior to viral fusion to ACE2 for replication. For SARS-CoV-2 and several other coronaviruses, these N-glycans serve as appendages for the virus to make its initial attachments to glycoconjugates on the host cell surface. These N-glycans, several of which are capped with terminal sialic acid (SA) moieties, sweep back and forth across spike protein like windshield wipers, partially shielding it from antibody binding. Key to the infectivity and morbidity of SARS-CoV-2 are glycans that protrude tangentially from 22 N-linked glycosylation sites on each monomer of its spike protein.  
They furthermore suggest therapeutic options using competitive glycan-binding agents such as IVM and may help elucidate rare serious adverse effects (AEs) associated with COVID-19 mRNA vaccines, which use spike protein as the generated antigen. These results validate and extend prior findings on the role of glycan bindings of viral spike protein in COVID-19. IVM blocked HA when added to RBCs prior to spike protein and reversed HA when added afterward. Omicron induced HA at a significantly lower threshold concentration of spike protein than the three prior lineages and was much more electropositive on its central spike protein region. The results of these experiments were, first, that spike protein from these four lineages of SARS-CoV-2 induced HA. Inhibition of spike protein-induced HA was tested using the macrocyclic lactone ivermectin (IVM), which is indicated to bind strongly to SARS-CoV-2 spike protein glycan sites. The electrostatic potential of the central region of spike protein from these four lineages was studied through molecular modeling simulations. To provide further insight into these glycan attachments and their potential clinical relevance, the classic hemagglutination (HA) assay was applied using spike protein from the Wuhan, Alpha, Delta and Omicron B.1.1.529 lineages of SARS-CoV-2 mixed with human RBCs. Experimental findings for SARS-CoV-2 related to the glycan biochemistry of coronaviruses indicate that attachments from spike protein to glycoconjugates on the surfaces of red blood cells (RBCs), other blood cells and endothelial cells are key to the infectivity and morbidity of COVID-19. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
